|
Established in 2001, CU Medical Systems covers the research, development, manufacturing and customer service of basic medical devices of defibrillators, utilizing not only the latest IT technology, but also its independently developed medical device technology and leading the domestic market over the past decade.
The company is domestically enjoying strong sales and has been exporting to about 70 countries around the world since 2001. The company is the only company in the nation covering a full range of defibrillators from AED for the public places to Defibrillator/Monitor for the hospitals.
The products of the CU Medical Systemsare are designed and manufactured according to international standards. All of its products have acquired many certifications such as ISO, CE, Korean FDA, USA FDA and SFDA.
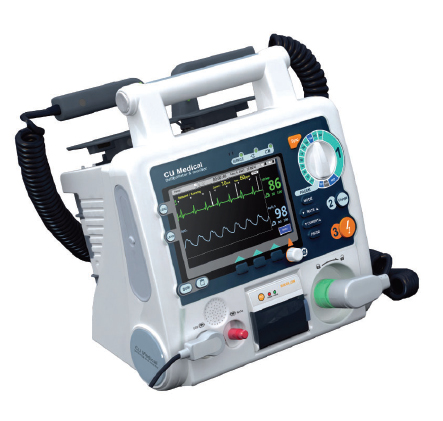
Among the company¡¯s various models of defibrillators, i PAD CU-SP1 is a relatively newly released defibrillator model that supports public access.
Providing built-in smart volume control system according to environmental noise, the model is also automatically capable of checking validity of pads. Informative LCD screen monitoring the status of pads and battery and displaying alarm is basically
provided. Pre-connected pads are supplied. Two kinds of battery options (DC 12 volt 2.8Ah, 4.2Ah) are available.
Another competitive model of i-PAD NF1200 is a semi-automated external defibrillator (also known as an AED). The i-PAD NF1200 is designed for minimally trained individuals. It provides simple and direct voice prompts and instructions for straightforward rescue operation. It is lightweight and battery powered for maximum portability.
The i-PAD NF1200 is intended to treat Ventricular Fibrillation (VF) and Fast Ventricular Tachycardia. These two conditions are the most common causes of sudden cardiac arrest (SCA). In SCA, the heart of the victim suddenly stops pumping. This condition may occur suddenly to any age group without warning. The only effective treatment for SCA is the application of a defibrillating shock.
|